- 本文轉載自《领研网》。撰文/ 戚译引 魏潇
編按:英國科學家於本月五日宣布 HIV 感染者「倫敦病人」在接受造血幹細胞移植後,血液中 HIV 病毒感染跡象完全消失,為全球第二例功能性治癒愛滋病患者。而後在六日,荷蘭科學家也在西雅圖愛滋病毒會議(CROI)宣布第三位在停藥後3個月也未在體內發現 HIV病毒的「杜塞道夫病人」( Düsseldorf patient)。他們是接受了怎樣的治療?愛滋病能夠痊癒已經是現在進行式了嗎?
日前(3/5),英國科學家宣佈一位 HIV 感染者在接受造血幹細胞移植後,血液中 HIV 病毒感染跡象完全消失,目前已經停藥 18 個月。《相關研究以letter形式於3月6日線上發表於Nature。》
這位患者被稱為「倫敦病人」,是科學家對「柏林病人」案例的首次成功複製。兩名患者都通過骨髓造血幹細胞移植獲得了攜帶 CCR5 突變基因的免疫細胞,這種方式是否能讓治癒 HIV 成為可能?或者說,CCR5 會成為人類戰勝 HIV 的突破口嗎?

2007 年,在西雅圖舉辦的逆轉錄病毒與機會性感染會議(Conference on Retroviruses and Opportunistic Infections)上,一位德國科學家報告了首例 HIV 感染者被功能性治癒的病例,這就是後來廣為人知的「柏林病人」。
時隔 12 年,在同樣的會議上,學界終於要迎來第二個同樣的病例——「倫敦病人」,他已經停用抗逆轉錄病毒藥物超過 18 個月,沒再出現 HIV 病毒感染跡象。
據相關媒體報導和論文報告,這名病人於 2003 年感染 HIV-1 病毒,2012 年確診血液癌症(霍奇金淋巴瘤);到 2016 年,患者病情已經十分嚴重,醫療團隊決定讓他接受造血幹細胞移植。手術後,患者只出現了輕微的排異反應,16 個月後抗逆轉錄病毒治療中斷。又過了 18 個月,患者血液中檢測不到任何 HIV-1 病毒的遺傳物質,研究人員將其稱為「功能性治癒」或「持續緩解」——這樣的案例太少,目前誰也不知道該如何定義「治癒」。
這項研究由來自英國、西班牙、荷蘭和新加坡的科學家合作完成,相關論文將於北京時間 3月6日淩晨在 Nature 網站上線。第一作者拉文德拉‧古普塔(Ravindra Gupta)目前就職於劍橋大學(Cambridge University),相關研究在倫敦大學學院(University College London)完成。還有一名病人已經停藥 4 個月,研究人員正在對其進行密切觀察。
CCR5 突變:攻破 HIV 的「銀色子彈」?
和「柏林病人」一樣,「倫敦病人」症狀緩解的關鍵在於一個重要的基因,CCR5。為了治療血液疾病,他們接受了骨髓造血幹細胞移植治療,進而擁有了一種罕見的基因突變,獲得了抵抗 HIV 病毒感染的能力。
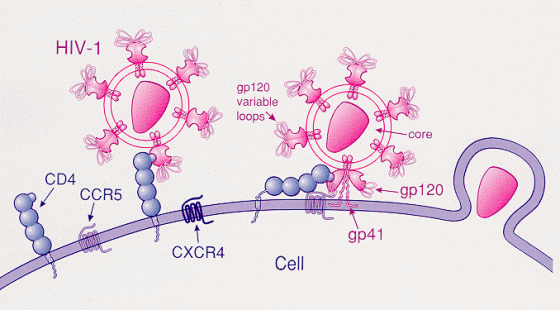
早在 1996 年就有研究者發現,CCR5 基因編碼的蛋白是 HIV 病毒入侵人體的「順風車」。CCR5 蛋白位於 CD4+ 輔助性 T 細胞的表面,像一個「小抓子」, HIV 病毒會利用它入侵輔助性 T 細胞,進而破壞整個免疫系統。更重要的是,研究人員發現一小部分人的兩個 CCR5 基因拷貝都產生了 CCR5 Δ32 突變,也就是說缺失了 32 個核苷酸,這讓他們表達的 CCR5 蛋白比正常情況下要短,不能到達 T 細胞表面,因此他們的 T 細胞對 HIV 感染有高度抵抗力。大約 1% 的高加索人遺傳了這樣的一對基因,但是在亞洲人、非洲人、美洲土著人中,這種基因非常罕見。
這次移植到「倫敦病人」體內的造血幹細胞就攜帶兩個 CCR5 Δ32 突變拷貝,保護輔助性 T 細胞免於 HIV 病毒感染。對於整個免疫系統來說,CD4+ 輔助性 T 細胞一旦能夠正常行使生理功能,就可以募集足夠的殺手 T 細胞,清除 HIV 病毒。
從上世紀九十年代起,多個研究組就將注意力放在了保護輔助性 T 細胞等免疫細胞不被 HIV 侵染的環節上,但直到二十一世紀臨床試驗的結果仍不理想。這時,「柏林病人」出現了。
史上第一例因骨髓移植「治癒」HIV的柏林病人
居住在德國柏林的 Timothy Ray Brown 1995 年被確診為 HIV 感染者,隨後一直服用抗逆轉錄病毒藥物控制病情,效果還算不錯。但是,2006 年他又患上了一種與 HIV 感染不相干的疾病——急性骨髓性白血病(acute myeloid leukemia)。Timothy 接受了化療,但白血病再次復發,為了活下去,他只能選擇利用他人的骨髓幹細胞在自己體內重建一個免疫系統。
骨髓移植中,捐獻者和接受者的人類白細胞抗原(human leukocyte antigen, HLA)配型一致非常重要,是保證患者不會出現免疫排斥反應的基礎。但是負責醫生傑羅·胡特(Gero Hutter)並不滿足於為 Timothy 找到一份僅僅是 HLA 匹配的骨髓幹細胞——他希望捐獻者同時還攜帶一對 CCR5 Δ32 突變的基因,讓 Timothy 利用這次「換血」的機會,獲得一個可以持續抵擋 HIV 入侵的全新免疫系統。
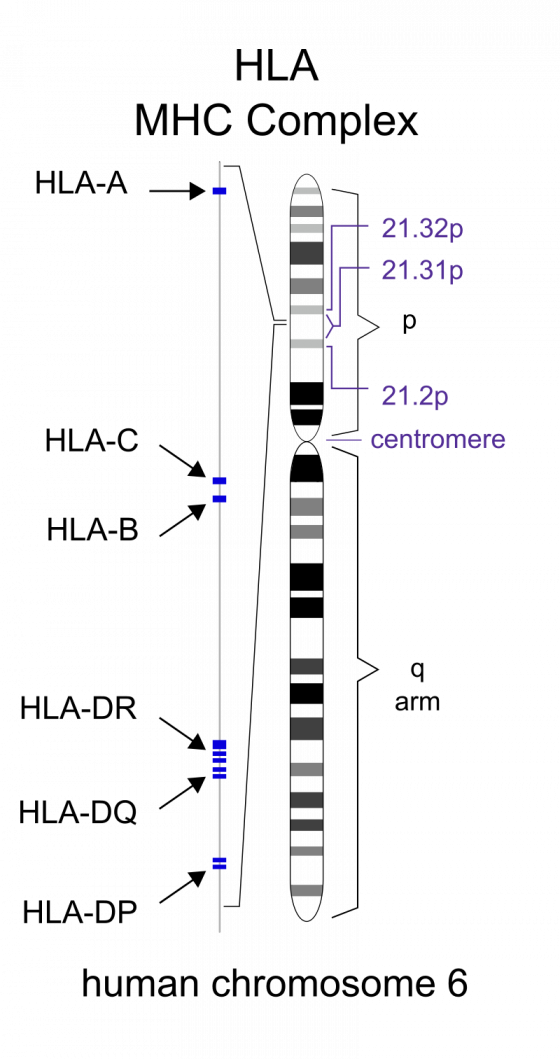
這是個極為複雜的搜索過程,因為 HLA 變化多端,人與人之間通常不一樣,而且 HLA 基因與 CCR5 基因位於不同的染色體。但令人驚訝的是,胡特成功找到了一個完全符合條件的捐贈者。
兩次骨髓移植後,Timothy 的白血病被治好了,同時,他在手術結束後再也沒有服用抗逆轉錄病毒藥物。從 2007 年手術結束到現在,醫生在他的血液、肝臟、腸道、大腦、淋巴組織及血漿中都沒有檢測到 HIV 病毒。Timothy 自此成為世界首個感染 HIV 後被「治癒」的患者,為了保護他的隱私,學界給了他一個「柏林病人」的代號,Timothy 在 2010 年主動公開了自己的名字,希望能鼓勵 HIV 感染者。
Timothy 的 HLA 配型較為常見,這可能稍微降低了一些難度。但是擁有如此精確匹配條件的捐獻者十分罕見,而且即便移植成功治好血液癌症,HIV 病毒卻不一定能得到控制。在這次的「倫敦病人」出現之前,沒人能重複傑羅·胡特的成功。
還不能確定被「徹底根治」
目前,沒人知道「柏林病人」是不是被「徹底根治」(sterilizing cured)了,因為 HIV 可以把自己的基因插入各種細胞的染色體中,但我們無法檢測,先前也有過病毒從血液中消失後又捲土重來的病例。至於這位「倫敦病人」,目前醫生只能確定他血液中的 HIV 病毒消失了,但其餘組織的情況 2 年後才能確定。
對於大多數 HIV 感染者來說,只要服用抗 HIV 藥物,就可以維持一種與普通人接近的生活(藥物的副作用和壽命的縮短仍然是無法回避的問題)。像柏林病人和倫敦病人那樣接受骨髓移植,並終身服用抗免疫排斥藥物,仍然是個風險極高的選擇。
成功案例能否再複製?
那麼,相同的治療方法是否有希望應用於中國的 HIV 感染者?
首先,CCR5 突變不能阻止所有 HIV 病毒。雖然 CCR5 蛋白是 HIV 入侵細胞的鑰匙,但它並不是唯一的那一把。HIV 存在的不同種類的病毒株,各有各的偏好:
- 嗜巨噬細胞的 HIV 病毒株會和巨噬細胞上的 CCR5 結合( R5 嗜性);
- 嗜 T 細胞的 HIV 病毒株會和 T 細胞上的 CXCR4 結合( X4 嗜性);
- 還有少數病毒株能和兩種共受體結合(雙嗜性)。
這次的「倫敦病人」感染的病毒全為 R5 嗜性,這很可能是移植了 CCR5 Δ32 突變的造血幹細胞後,病人血液中 HIV 病毒被完全清除的重要原因之一。但是,HIV 的表面蛋白 gp120 突變率很高,當 CCR5 這條路走不通時,它能很快採用備選方案:和 CXCR4 結合——我們不能輕易忽視這種可能。
另外,在中國流行率最高的 HIV 病毒株為 HIV-1 CRF01_AE,其中呈 X4 嗜性的比例顯著高於其他亞型:一項廣西佇列研究(cohort study,另可譯為世代研究、分群研究)顯示,約 61% 的HIV帶原者攜帶 HIV-1 CRF01_AE,而另一項上海佇列研究發現這種病毒株中有 32.4-40.9% 能通過和 CXCR4 結合進入細胞。也就是說,針對 CCR5 的預防或治療手段可能對中國的很大一部分 HIV 病毒無效。
其次,CCR5 的缺失可能帶來未知風險。CCR5 雖然為 HIV 病毒大開方便之門,但它並非一無是處。事實上,CCR5 在巨噬細胞和 T 細胞的交互作用中扮演重要角色,CCR5 缺失很可能造成機體免疫應答紊亂,對某些感染抵抗力更差,並影響腫瘤的發生和治療。已有多篇研究報導,CCR5 Δ32 突變個體面對多種感染性疾病時的預後較差,例如感染流感病毒的死亡率是對照組的 4 倍。還有研究顯示,CCR5 表達缺失會導致癌症免疫療法效果較對照組差。
還有觀點認為 CCR5 與認知功能有關。近期發表在 Cell 上的一篇文章發現,CCR5 缺失和CCR5 Δ32 突變小鼠在腦損傷後,恢復學習能力及認知記憶的能力較佳。但需要注意的是,此類研究還不能說明 CCR5 缺失或突變能夠改善認知功能。
總而言之,從目前的兩個病例看來,CCR5 或許可以成為一個重要的突破口,為部分 HIV 感染者帶來新的希望,但是那些感染其他毒株的患者還迫切需要新的治療手段。而對於健康人而言,CCR5 在免疫系統乃至整個機體中發揮著重要的作用,對它的缺失可能帶來的風險和收益,我們還無法得出明確的答案。
- 迦陵頻伽(巴黎第五大學免疫學博士)對此文亦有貢獻。
參考文獻
- Gupta, R. K., Abdul-jawad, S., McCoy, L. E., Mok, H. P., Peppa, D., Salgado, M., … & Grant, P. (2019). HIV-1 remission following CCR5Δ32/Δ32 haematopoietic stem-cell transplantation. Nature, 1.
- 《HIV Circulating Recombinant Forms (CRFs)》
- AlkhatibG, The biology of CCR5 and CXCR4. (2010) CurrOpin HIV AIDS.
- Lieberman-BlumSS, Fung HB, Bandres JC, Maraviroc: a CCR5-receptor antagonist for thetreatment of HIV-1 infection. (2008) ClinicalTherapeutics.
- AndersonJ, Akkina R, Complete knockdown of CCR5 by lentiviral vector-expressed siRNAsand protection of transgenic macrophages against HIV-1 infection. (2007) Gene Therapy
- Li J, et al., HIV-1 Transmissions AmongRecently Infected Individuals in Southwest China are Predominantly Derived fromCirculating Local Strains. (2018) ScientificReports
- Li X, et al., Evidence That HIV-1 CRF01_AE Is Associated with Low CD4+T Cell Count andCXCR4 Co-Receptor Usage in Recently Infected Young Men Who Have Sex with Men(MSM) in Shanghai, China. (2014) PLOS One
- Klein RS, A Moving Target: The Multiple Roles of CCR5 in Infectious Diseases. (2008) J. Infect. Dis.
- Glass WG et al., CCR5 deficiency increases risk ofsymptomatic West Nile virus infection. (2006) J. Exp. Med.
- Ahlenstiel G et al., Effects of the CCR5-Delta32mutation on hepatitis C virus-specific immune responses in patients withhaemophilia. (2009) Immunol. Invest.
- González-Martín A et al., CCR5 in cancer immunotherapy. (2012) Oncoimmunology
- Vangelista L and Vento S, The Expanding Therapeutic Perspective of CCR5 Blockade. (2017) Front.Immunol.
- Joy MT et al., CCR5 Is a Therapeutic Target forRecovery after Stroke and Traumatic Brain Injury. (2019) Cell
- 《Has a second person with HIV been cured?》
- 關閉HIV入侵之門,《環球科學》2012年第4期
- 《Tests on London patient offer hope of HIV ‘cure’》
- 《H.I.V. Is Reported Cured in a Second Patient, a Milestone in the Global AIDS Epidemic》
- 本文原文為〈Nature | 全球第二例或被治愈的艾滋病患者:体内HIV被清除,已停药超18个月〉未經許可請勿轉載,獲取授權請聯繫《contact@linkresearcher.com》。