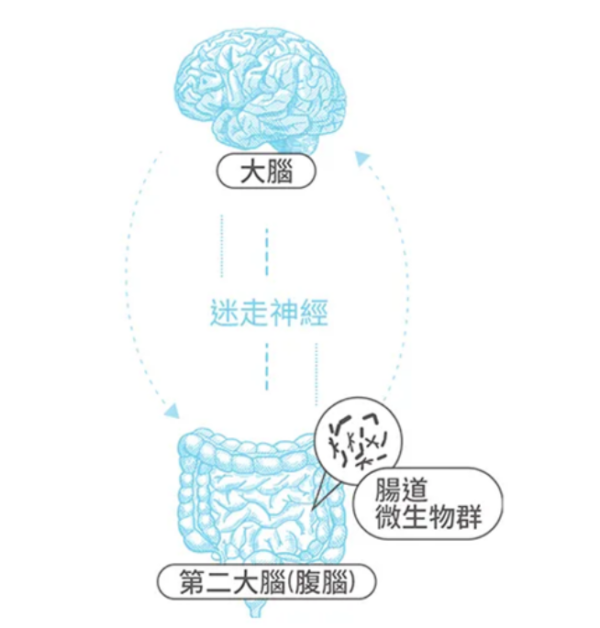
看到 「人帥益生菌,人醜大腸菌」這句話在 FB 上洗板,細菌人眉頭一皺,因為這句話乍聽之下覺得有趣又有點科味,但覺得有點怪怪的。到底是哪裡怪怪的呢?
細菌絶不會加入外貌協會
這個人帥人醜居然也可以牽拖到細菌,不知道細菌是最不重視外在,還是最不在外形上講究的族群嗎?想看到細菌的臉長什麼樣,一般的顯微鏡還辦不到,得動用到電子顯微鏡放大個數上萬倍才可以。但放大以後的細菌長得帥嗎?跟你講個秘密,大部份的細菌在掃描式電子顯微鏡下,要不長得像嘟嘟好香腸(短桿菌),要不就像一粒粒的魚丸(球菌),每隻都長那個樣,老實說看了挺無趣的(或看了挺餓的)。益生菌或是大腸菌,長得都差不多,細菌是絶不會加入外貌協會的!
所以這句話是要說人帥人人愛,人醜就心眼壞了囉?要能這樣講,你心裡大概默認益生菌是好東西,大腸菌則是可怕討厭的壞東西。不過,真的是這樣的嗎?
散播益生菌散播愛
益生菌最早是二十世紀初期荷蘭科學家埃黎耶.梅契尼可夫(Élie Metchnikoff)所提的概念,指的是吃了能有益人類健康的微生物。所以就像很多廠牌型號的通訊機器,不管有多少功能,統統稱作手機一樣,益生菌指的是任何一種符合這項標準的微生物。
我們的身體裡有益生菌嗎?拿兩類最常見的益生菌乳酸桿菌(Lactobacillus)和比菲德氏菌(Bifidobacterium)來說好了,這兩類細菌在身體裡都有,前者在小腸環境多,後者在大腸環境多,但數量會變動,如果能由體外補充或許不錯。
那,我們會到處散佈益生菌嗎?這件事就不容易發生了。乳酸菌雖然在小腸數量多,可是在通過腸道後數量變少,在環境裡也不容易存活,只佔糞便總菌量的一小部份。而比菲德氏菌是厭氧細菌,在有氧氣的環境下無法生存。要從人身上得到這些菌實在是很困難,你大概得每天接受那人的接種好久好久,才有可能成功接到一次。除了一種特殊狀況:女性陰道裡有不少乳酸桿菌,我們在出生時從那裡帶走了第一批保護自己的細菌。不過這種機會一輩子也就只有那麼一次了。
益生菌,你愛不愛?
印象中的益生菌都是要花錢買的。常常有人問我,益生菌吃了真的有效嗎?我也常常回答,這要看你的體質,別問我我不知道耶(然後得到白眼一雙)。
證明某益生菌有效的研究有好多好多,你可能也聽過某伯伯家的小孩吃了益生菌後異位性皮膚炎變好了,但如果真要保證在你身上一定有效,則有點困難。常被用來當做益生菌的乳酸桿菌(Lactobacillus)的菌種,就對我們的黏膜免疫系統(mucosal immune system)做了不少微調的動作。我們吃益生菌,某個程度上等於是拿菌當藥來微調我們自己的免疫系統。但是每個人的免疫系統對什麼東西容易發生反應並不太一樣,能微調阿伯家小孩的菌不見得調得動你的系統,或許,微調的方向不見得是對你有用的。就像每天早上廣播裡出現的升學教材廣告一樣,同樣的教材給你,你唸了就保證能達成你的升學夢嗎?有的人可能就無法喜歡教材裡的編排方式呢!
益生菌對你可能有效可能無效,但是花錢買了才能試,這點大概大家都一樣。
叫大腸桿菌卻不愛大腸
大腸菌指的應該是大腸桿菌吧!大腸桿菌顯然住在臭臭髒髒的大腸裡,連政府都會檢驗食物飲料裡大腸桿菌的數量,數量超標了就有食安顧慮要開罰的。這種細菌,總不可能會是個有人喜歡的傢伙了吧?
你錯了。大腸桿菌不愛大腸,當初它是從健康嬰兒的糞便裡被找到的,糞便來自大腸,所以得到這名字。但其實大腸桿菌比較喜歡住在小腸裡面,因為小腸裡養份多,而且還有一些氧氣。到了大腸氧氣耗盡,這個沒有氧氣的世界裡住滿了各式各樣的厭氧菌。大腸中有很多人類消化不了的植物纖維,準備貼上大便標籤後被丟掉,因此很多大腸住民具有切斷植物纖維來當食物的本領,才能在這裡討生活。大腸桿菌啃不了植物纖維,在這裡混不下去,只能撿人家掉的屑屑為食,其實數量在這裡是多不起來的。
每個人都有大腸桿菌
在大家眼裡,大腸桿菌跟益生菌最大的不同,應該是你得花錢買益生菌,但是每個人的肚子裡都配備了自己的大腸桿菌。大腸桿菌早就適應在我們的小腸裡的生活,也在小腸裡成為優勢菌群。
我們讓這些不作怪的大腸桿菌住在身體裡其實是有好處的,因為當病原菌從你吃的食物裡通過胃進到小腸,如果想要在小腸定居致病,第一步是要先打敗大腸桿菌這些地頭蛇,才能佔到立足之地。如果你剛好吃了抗生素把這些常駐的居民清除掉,受感染的機會可以要變高很多。另外大腸桿菌也能製造人類需要攝取的維生素 K,在腸子裡養這種細菌應該是有點好處的,只是我還沒找到直接證據證明人真的從大腸桿菌那裡得到這養份。
看起來大腸桿菌好像應該是我們的朋友。那為什麼電視上會說,某家飲料的大腸桿菌數超標不合格,那它不就應該是個好可怕的有害細菌嗎?這又要好好說明一下了。
這種測試背後的想法來自於大腸桿菌是人的腸子裡才會出現的細菌,而且在環境中活不了太久就會死掉。所以當飲水或食物中出現它,那表示沒多久前還活在某人、某動物腸子裡的東西,和這些飲水或食物有接觸。雖然大腸桿菌可能對我們無害,但是它都可以出現,萬一它的源頭是個散佈病菌的病人,在病人腸子裡的病原菌就有機會進到你的食物或飲水裡,很不妥。2010 海地的霍亂大流行就是這樣來的。
大腸桿菌殺人事件
聽起來好像大腸桿菌是好房客,又會禦敵又能生產有用的養份。不過,大腸桿菌也能在環境裡居住,而且會在撿到致病基因後重新回到舒適的腸道老窩。這時就不好玩了,這些大腸桿菌不再安份當個房客,會進一步攻入人體組織。其中在腸道裡危害嚴重的有:產毒性大腸桿菌(enterotoxigenic E. coli)、致病性大腸桿菌(enteropathogenic E. coli)、侵入性大腸桿菌(enteroinvasive E. coli)、出血性大腸桿菌(enterohemorrhagic E. coli)等等。不只腸道,泌尿道致病性大腸桿菌(Uropathogenic E. coli)還會從尿道進攻,在又酸又毒的尿液裡逆流而上,甚至攻進腎臟。這些病原,每年奪走好多人的性命,光是死在產毒性大腸桿菌手上的人就有數十萬。殺人兇手無誤。
雖然這些特殊菌株有感染人的能力,但是絶大部份的大腸桿菌是無害的,要把少數極端份子的帳算在大腸桿菌上,看起來也不是那麼公道的啦。
結論
人帥人醜都一樣,在正常的狀況下,你散播下的都不太可能是益生菌,但留下大腸桿菌的機會是有的。人或動物留下的大腸桿菌造成重大危害的機會不大,但是大腸桿菌是個指標,告訴你大便來了。而不管你是誰,在你腸子裡的益生菌和大腸桿菌還是會繼續保護你的。