今年十月,駐守在國際太空站的法國太空人 Thomas Pesquet 拍攝到難得一見的異象:耀眼的藍白色光暈在歐洲大陸上空炸開,彷彿《天使與魔鬼》中的反物質已經被引爆。
🌩A single frame from a timelapse over #Europe, showing a transient luminous event in the upper atmosphere! We have a 🇩🇰-led facility monitoring these events thanks in part to @Astro_Andreas who took the first picture of them from space! ⚡ https://t.co/tfFS3KqYmm #MissionAlpha pic.twitter.com/XqBdJ64pBq
— Thomas Pesquet (@Thom_astro) October 7, 2021
不過這陣閃光並沒有登上當地新聞,甚至沒幾個人看到他發生。因為它並不是發生在地面上的事件,而是大氣層上層的放電現象,稱為瞬態發光事件(Transient Luminous Event, TLE)。
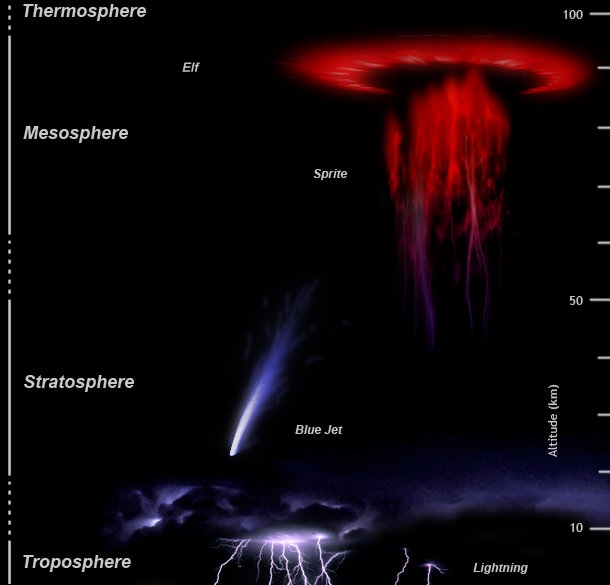
往上打的雷
如同伴隨午後雷陣雨而來的閃電與雷聲,這些發光事件源自於雲層中充沛的電荷。在午後雷陣雨時,雷雨雲中的水滴與冰塊在劇烈的上升氣流中摩擦生電,使得雲層頂部和底部累積許多焦躁的電荷。一旦電荷累積足夠,巨大的電位差便會往地面打通一條游離化的導電通道,進而在陰雨的天空中劈出一道亮光。這就是我們常見到的雲對地閃電。
不過有時候,雲層中的電荷如果率先找到了向上的通道,便會產生「向上」的閃電,稱為「藍色噴流(Blue Jet)」,屬於 TLE 的一種。由於成因和一般雲對地閃電類似,這種宛如外星傳送門一般的景象有時被稱為中高層大氣閃電,但藍色噴流聽起來酷多了。雖然拍攝者沒有特別註明,不過這次在國際太空站拍攝到的有可能是藍色啟動器(Blue Starter),也就是長度較短的藍色噴流。
雲層上方的平流層和中氣層也時常見到另一種 TLE,叫做紅色精靈(Red SPRITE)。這種橘紅色的光影是受到下方閃電擾動後產生的高層放電所導致。他們通常成群出現,有著各種五花八門的型態,一般以下方觸鬚形狀分為水母狀和紅蘿蔔狀兩種。更高的電離層則有暗紅色的淘氣精靈(ELVES)。

大氣科學家的小幽默
到這裡你可能已經發現,科學家為這類現象命名的品味十分獨特,而且他們還花時間將這些有趣的名字硬是編成煞有其事的縮寫。SPRITE 是強烈雷雨雲電離導致的平流層擾動(Stratospheric/mesospheric Perturbations Resulting from Intense Thunderstorm Electrification);ELVES 則是電磁脈衝源造成的甚低頻擾動與發光現象(Emission of Light and Very Low Frequency perturbations due to Electromagnetic Pulse Sources)。為了延續這個主題,後來陸續被觀察到的現象也以這類概念取名,包括山怪巨魔(TROLL)、小妖精(Pixies)、幽靈(GHOST),和地精(Gnomes),為大氣科學增添許多奇幻色彩。
觀測紀錄太少,一度被視為謠言
這些曇花一現的大氣奇景過去只是飛行員間口耳相傳的傳說,被學界斥為無稽之談。一直到 1989 年,藍色噴流才首次被太空梭拍攝的黑白影片捕捉,證實了這些傳說中的閃光確實存在。在觀測影像技術進步的同時,人們也開始積極尋找觀測高層大氣的放電現象。世界各地的照片與錄影時不時紀錄到各種不同顏色和型態的 TLE。不過由於這些奇幻的光芒只存活在雲層上方的高空,而且通常只維持幾毫秒,要從地表觀察很碰運氣,使得至今關於這類現象的資料量仍然不多。
如果想要做觀測,除了像飛行員一樣在高空中拚巧遇,最好的方法就是從太空攝影。因此,國際太空站便成為 TLE 的最佳觀測站。這已經不是第一次駐紮上方的太空人目擊類似的現象,只要打開縮時攝影,就能捕捉這些轉瞬即逝的景象。由於赤道附近有許多雷雨雲,每當太空站經過這個區域時便是最佳觀察時機。
除了地球之外,NASA 去年也透過朱諾號探測器在木星大氣層拍到類似精靈的放電發光現象。只不過這些精靈跟地球上的紅色精靈有些差別。紅色精靈的色彩來自於地球大氣中豐富的氮氣成分,不過木星大氣主要由氫氣組成,所以出現了前所未見的外星藍色精靈。科學家以前就預測:在木星這類同樣有大氣和閃電的星球上也能見到 TLE,不過一直到去年才首次直接觀察到,再度顯示這類觀測研究的難度。
根據福爾摩沙衛星二號的觀測資料,科學家估計全球每年會發生數百萬次 TLE。關於它們的成因和物理機制,目前只能算是略懂略懂,而其對氣候和高空飛行器的影響也有待科學家繼續探索瞭解。目前國際太空站上的哥倫布號實驗艙外已經配備專門的設施,針對 TLE 進行監控拍攝。將來或許能見到更多絢爛的奇幻物種現身在遙遠的空中。
參考資料:
- What The Heck Was This Blue ‘Luminous Event’ Photographed From The Space Station?
- Andreas Mogensen captures gigantic lightning from the International Space Station
- Lightning ‘Sprites’ Have Been Detected on Jupiter For The First Time, NASA Says