在上一篇文章我們介紹了闊胸波溫蘇鐵象鼻蟲 (Miltotranes prosternalis) 的生態習性(見一生只為一人傳情,卻被誤解了 16 年的波溫蘇鐵象鼻蟲),也帶大家認識了蘇鐵這類外型類似棕櫚的熱帶、亞熱帶木本裸子植物,它們有著經濟重要性,可被作為田園造景植物,也身居保育價值而名列於國際自然保護聯盟 IUCN 的保育名錄和《瀕臨絕種野生動植物國際貿易公約》 (CITES) 附錄。
雖然蘇鐵含有有毒物質──蘇鐵苷,會造成肝腸胃道疾病以及具備神經毒性,然而世界一些地方的原住民,仍然會將蘇鐵樹幹磨粉後,再進行反覆淘洗等工序來去除毒性以供食用,日本奄美群島的居民甚至會利用琉球蘇鐵的種子來製作「蘇鐵味噌」。
而在人工栽培的蘇鐵身上,卻有著讓園藝栽植者氣到大喊:「有沒有學生要來研究怎麼防治牠呀?」並公開呼籲設立專項研究生獎學金的澳洲蘇鐵蛀莖象鼻蟲,這到底是怎麼一回事呢?

蛀食蘇鐵莖幹的澳洲象鼻蟲
蘇鐵褐蛀象鼻蟲 (Demyrsus) 和蘇鐵黑蛀象鼻蟲 (Siraton) 是僅分佈於澳洲的象鼻蟲特有屬別,幼蟲會蛀食蘇鐵的莖幹,這兩個屬別的象鼻蟲在澳洲的天然寄主植物是鱗木澤米蘇鐵 (Lepidozamia) 及大澤米蘇鐵 (Macrozamia) 兩類蘇鐵。
在野外,牠們僅會侵襲死亡或生病不健康的個體,並不會造成危害,然而在人工環境如植物園、花園和苗圃場內,牠們被發現除了澳洲蘇鐵外,他們還能攻擊外國產的蘇鐵類群,並造成大量死亡,有曾經意外被引入美國、義大利、比利時和南非的紀錄,甚至當初蘇鐵黑蛀象鼻蟲這個屬被發現時,就是以在義大利採集的標本所發表描述的,事隔多年人們才發現牠並不產於義大利而是遙遠的澳大利亞。



蘇鐵是一種生長速度非常慢的植物,所以可想而知栽種者大半輩子的心血付之一炬的痛心,甚至曾經有位蘇鐵愛好者在園藝雜誌上呼籲設立一個研究生獎學金機會來資助防治這種象鼻蟲,可見得該苦主有多麼悲憤。
由於這些象鼻蟲蛀食的位置非常深而且蘇鐵莖幹質地又非常堅硬,當園主發現後院的蘇鐵盆栽漸漸落葉,並且有不明的甲蟲鑽出時,感染狀況早已病入膏肓,而這些特性也造成現行的藥劑施用仍無能為力,所以目前僅能靠限制大型野生植株進口和檢疫來管控,以期能減少意外攜入這些「澳客」的機會,國際自然保護聯盟 IUCN 蘇鐵專家群已經將這兩類象鼻蟲都列為最高威脅層級的蘇鐵害蟲,幸好目前都是零星的發現紀錄,尚且沒有在國外長期立足的狀況。
澳洲蘇鐵蛀莖象鼻蟲的分類學研究
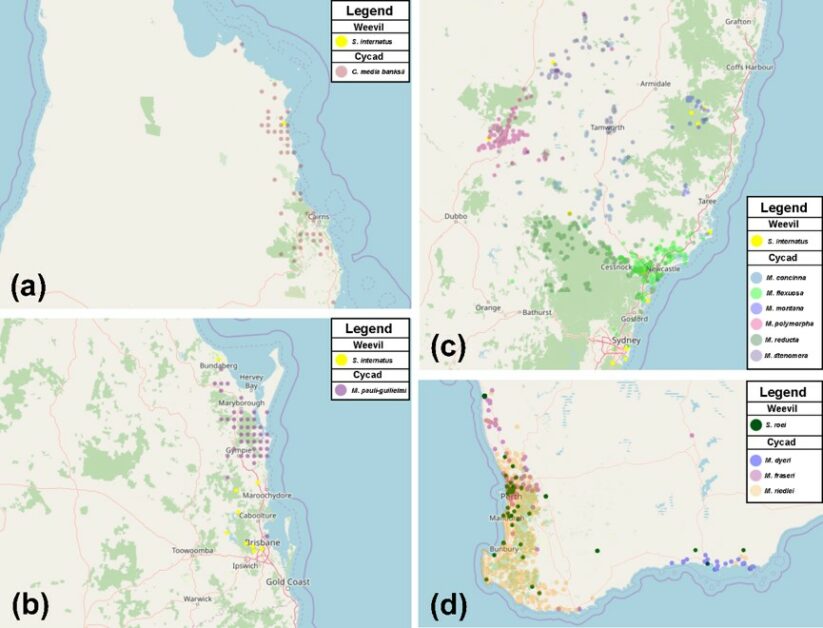
在我們 2020 年底發表於澳洲昆蟲學會的學術期刊「南方昆蟲學 Austral Entomology」的論文中 (Hsiao & Oberprieler, 2020),我們研究了蘇鐵褐蛀象鼻蟲和蘇鐵黑蛀象鼻蟲的分類學,我們檢視了成蟲、蛹和幼蟲形態,重新釐清和定義了屬級和種級的特徵,整理了完整的物種形態鑑定資料,並提供了成蟲和終齡鑑定檢索表和地理分佈圖,在我們的研究過程中,除了已知種的蘇鐵褐蛀象鼻蟲 (D. meleoides)、中間蘇鐵黑蛀象鼻蟲 (S. internatus)和羅氏蘇鐵黑蛀象鼻蟲 (S. roei)。
我們驚訝地發現產於北昆士蘭的蘇鐵褐蛀象鼻蟲是一個尚未被世人發現的全新物種,我們將新種象鼻蟲命名為鋼鑽蘇鐵褐蛀象鼻蟲 (D. digmon),學名源自日本經典卡通「數碼寶貝大冒險 02」中的裝甲體昆蟲型數碼獸「鋼鑽獸 ディグモン」命名,鋼鑽獸擁有操縱大地的力量,可旋轉鑽頭在地面給予沖擊以引起地割,跟我們這種鑽到蘇鐵莖幹深處害蘇鐵死亡的象鼻蟲非常地相似。

另外我們透過蘇鐵物種和象鼻蟲分佈的比較,也列出了數種潛在的蘇鐵寄主種類,可供有關檢疫單位參考,由於我們所發現的此新物種的標本籤上明確記載著會攻擊非洲特有的稀有蘇鐵──非洲蘇鐵屬 (Lepidozamia),所以很有可能也有潛在的危害性。
熟悉的蘇鐵最對味──澳洲蘇鐵蛀莖象鼻蟲的跨屬危害及演化啟示
如前文所述,蘇鐵褐蛀象鼻蟲和蘇鐵黑蛀象鼻蟲除了澳洲產的蘇鐵之外,還能危害外國產的蘇鐵,而在分析手邊的危害紀錄後,我們發現了有趣的生物學現象,雖然兩類象鼻蟲可以攻擊好幾屬外國的蘇鐵,然而根據統計,危害的記錄集中在非洲蘇鐵屬 (Encephalartos) 的物種上。
如果我們將這樣的寄主偏好連結上蘇鐵的演化樹,我們驚奇地發現非洲蘇鐵和作為天然寄主植物的澳洲鱗木澤米蘇鐵及大澤米蘇鐵是共同形成一個支序,是共享最近血緣的近親類群。
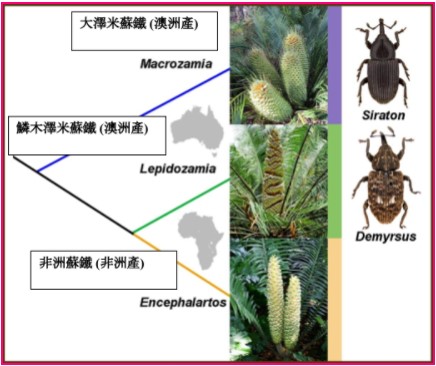
在演化上的近緣性說明了這些蘇鐵可能在生理上特性相近,所以當這些象鼻蟲意外的被帶到遙遠的異鄉國度,身為澳洲蘇鐵親戚的非洲蘇鐵因此對於這些象鼻蟲更好「入口」,可說是最熟悉的異國料理。
另外,由於澳洲和非洲從前都是從岡瓦那古大陸分離,所以這些象鼻蟲攝食起蘇鐵的起源也令人相當地好奇,到底這樣的食性是在岡瓦那古陸時期就已經演化生成,並且根據澳洲蘇鐵有著非洲蘇鐵遠親的鏡像性,澳洲蘇鐵蛀莖象鼻蟲在非洲也曾經有著類似的近親象鼻蟲兄弟但後來因不明原因滅絕,抑或是說這些象鼻蟲是正港澳洲出產的特有類群,牠們是澳大利亞自岡瓦那古大陸分離後才誕生演化出來,後天水平地獲得了這種「口味偏好」(土生土長的台灣人在習慣台式炸排骨後,某一天嘗到日式炸豬排後不可自拔?),雖然值得研究的細節還很多,但我們目前並沒有發現有任何證據支持前者的「兄弟滅絕說」。
總而言之,從演化生物學上的觀察我們得以呼籲園藝業者、愛好者和植物園經營方,如果你不想要你心愛的蘇鐵植株死亡,那麼你應該要避免引進野生的鱗木澤米蘇鐵及大澤米蘇鐵,並將其與非洲蘇鐵種植在一起。
參考文獻
Hsiao, Y. Oberprieler, R.G. 2020. A review of the trunk-boring cycad weevils in Australia, with description of a second species of Demyrsus Pascoe 1872 (Coleoptera: Curculionidae). Austral Entomology 59 (4): 677-700.