生活中總有那種到商店選購了很多商品,結帳後卻發現自己忘記攜帶環保袋的經驗,這時候銅板價的塑膠袋就是把商品輕鬆帶回家的好選擇。根據統計,每年全世界使用高達 5 億個一次性塑膠袋。
你知道塑膠袋發明是為了重複使用嗎?
瑞典工程師圖林 (Sten Gustaf Thulin) 發明了第一個塑膠袋。因為相較於紙袋,塑膠袋較不容易因為潮濕或拉扯破損,利於重複使用,能減少樹木砍伐,而且相對於紙袋,塑膠袋也較能攜帶重物。透過塑膠袋的發明,圖林希望解決紙袋造成的環境問題。不過統計至今,其中超過半數的塑膠袋並沒有達成「重複使用」這個使用初衷,有許多塑膠袋反而被隨意扔棄在大自然中,造成了另一種的生態壓力。
過去幾年來,塑膠製品嚴重影響環境生態。數年前,一支我們習以為常的吸管卡進海龜鼻子裡,短短 8 分多鐘的 Youtube 影片,驚醒了許多人的環保意識,開始拒用一次性塑膠吸管,行政院環保署也在 108 年 5 月宣布一次性塑膠吸管使用規範。除了限制一次性使用塑膠吸管,部分商店與機構也紛紛推出自備環保餐具即享折扣的優惠,對於不得不使用的一次性塑膠製品,政府也提倡回收再利用。
2018 年,台灣塑膠總回收量為 53 萬公噸,而其中寶特瓶回收量則高達 54 億支,資源回收率號稱全球第三,寶特瓶的回收量更高達 95%,即便是這樣,每年仍有 2-3 億支寶特瓶流入大自然,因此除了推行塑膠減量外,科學家也開始思考如何減短膠製品的分解週期。
發現會吃塑膠的細菌
塑膠製品那麼多,那就先從民生用品最常見的 PET(苯二甲酸乙酯)說起。
PET 是一種常見的塑膠材料,在衣服、包裝及地毯都可以看到它的蹤跡,其中 PET 最廣為人知的製品就是寶特瓶。PET 價格低、輕便的特性讓他成為常見的塑膠材料,不過卻也因為結構中將兩個單體聚合在一起的酯鍵並不容易被分解,因此有著驚人的抗生物分解能力,分解一支寶特瓶至少需要長達 450 年的時間。
即便數據顯示,目前寶特瓶的回收率並不低,但在台灣每年卻還是有 2-3 億支寶特瓶沒有成功回收。這些被遺忘的寶特瓶最終被丟棄在人類的生活軌跡,好一點的丟進垃圾掩埋場,運氣不好的則被帶入山林或流進海洋,再次造成野生動物們的傷害。

等等!剛才說分解寶特瓶需要 450 年,那就是還有辦法分解啊,所以只要找到這些年是誰在分解寶特瓶,事情不就好喬了?
沒錯!首先想到的就是來自日本的研究團隊,他們將寶特瓶回收場的底泥、濕土、廢水,只要是有接觸到分解寶特瓶殘骸的環境樣本通通帶回實驗室。研究團隊將這些帶回來的樣本培養在 PET 塑膠薄膜上,希望看看是不是有微生物在分解 PET,可以靠著分解 PET 產生能量,維持細胞中的代謝與合成反應。
結果真的有。
2016 年,日本研究團隊發現了一株愛「吃」塑膠的新型細菌,這株細菌可以在大概 6 週的時間內,將 PET 薄膜完全分解。研究團隊將它命名為大阪堺菌 (Ideonella sakaiensis)。
生物產生的催化劑——酵素
找到新細菌然後呢?想知道他可能有什麼神奇的超能力,最快的方法就是送去定序。
研究人員根據定序結果發現細菌帶有一種水解酵素,推測這種酵素可能與大阪堺菌能分解 PET 有關。為了證明這樣的推測,研究人員進行酵素純化,並測試其分解 PET 的能力,發現在攝氏 30 度的環境中,酵素有最佳的反應效率將 PET 分解,決定命名為 PETase。
團隊在找出大阪堺菌分解 PET 的關鍵機制後,成功以化學方法純化 PETase,擺脫微生物實驗中的許多條件限制。不過能在 6 週內分解 PET 塑膠薄膜只能確定酵素有分解的能力,但是真正要面對 2-3 億支比薄膜更厚實的 PET 寶特瓶,這樣的實驗成果遠遠不夠,因此科學家們紛紛開始思考如何加快分解 PET 的反應速率。
找到能分解塑膠的酵素,那能再快一點嗎?
2018 年英國樸茨茅斯大學 (University of Portsmouth) 研究團隊根據先前研究分析 PETase 的結構,發現其胺基酸序列與嗜高溫放線菌 (Thermobifida Fusca) 的水解酵素 TfH 有 52% 的相似度,但是兩者之間對於受質的專一性不同,其中 PETase 的結構相似於催化角質與脂肪酸分解的 α/β 水解酵素,因此研究團隊希望透過改變 PETase 的結構,設計出一種酵素能夠更快速分解 PET。
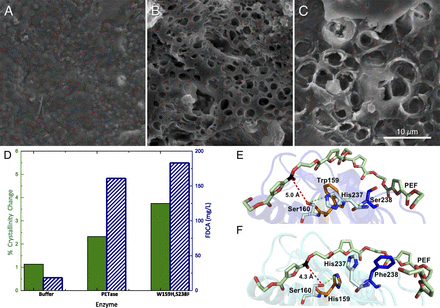
哈里·奧斯汀 (Harry P. Austin) 研究團隊將 PETase 的結構突變為更類似於此水解酵素,嘗試讓兩個水解酵素中的兩個活性位點,由殘基突變為保守氨基酸,使結合裂變窄,優化 PETase 對於 PET 的分解能力。在實驗中將野生型 PETase 和 PETase 雙突變體 (S238F / W159H) 透過掃描式電子顯微鏡進行比較(如圖 A-C),A 為緩衝溶液(對照組),B 為野生型 PETase,C 為 PETase 雙突變體 (S238F / W159H) ,在 pH7.2 的磷酸鹽緩衝溶液培養 96 小時後,可以看到對照組的 PET 表面沒有被分解產生的孔洞,而 PETase 雙突變體的孔洞較野生型 PETase 多且明顯,應證了雙突變的 PETase 在相同條件下,能更有效的分解 PET。
雖然透過科學家們的努力,在未來有機會利用蛋白質工程,嘗試更多組合,找到讓塑膠分解更快速的方法。不過以根本來說,降低塑膠使用量、在未來審慎考慮各種材料的利用,照顧好地球環境,才是我們的首要目標。
參考資料
- Bag with handle of weldable plastic material
- How plastic bags were supposed to help the planet – BBC News
- 從海龜氣管拔出塑膠吸管
- 環保署公告「一次用塑膠吸管限制使用對象及實施方式」
- 台灣成就!寶特瓶回收率高達95%、資源回收率全球第3 但真的夠了嗎?
- 塑膠垃圾別亂丟,海洋分解得花600年
- Yoshida, S., Hiraga, K., Takehana, T., Taniguchi, I., Yamaji, H., Maeda, Y., … & Oda, K. (2016). A bacterium that degrades and assimilates poly (ethylene terephthalate). Science, 351(6278), 1196-1199.
- Austin, H. P., Allen, M. D., Donohoe, B. S., Rorrer, N. A., Kearns, F. L., Silveira, R. L., … & Beckham, G. T. (2018). Characterization and engineering of a plastic-degrading aromatic polyesterase. Proceedings of the National Academy of Sciences, 115(19), E4350-E4357.